Greenhouse gases
Greenhouse gases are the main cause of Earth’s increasing temperature, an important part of climate change.
What are greenhouse gases?
Greenhouse gases absorb and emit radiation in the infrared range, which we feel as heat.
Short-wave radiation is emitted by the Sun and mostly passes through Earth’s atmosphere. The Earth’s surface absorbs most of this and re-emits it as infrared radiation at a longer wavelength.
Greenhouse gas molecules absorb some of this infrared radiation, instead of letting it pass straight out into space. The molecules then re-emit infrared radiation in all directions, including back to the surface. This causes the planet to heat up and is known as the 'greenhouse effect'.
Without greenhouse gases in the atmosphere, the Earth would be about 30 degrees colder, as all the infrared radiation from the surface would be able to escape into space. Greenhouse gases allow the Earth’s surface to be habitable.
Greenhouse gases are also found on other celestial bodies, including Venus, Mars, and Titan, a moon of Saturn. Indeed, greenhouse gases contribute to Venus having a surface temperature of over 400°C.
Greenhouse gas emissions
Emissions of greenhouse gases from human activities enhance the greenhouse effect. This increases the average temperature of the Earth and contributes to climate change.
Human greenhouse gas emissions primarily come from burning fossil fuels for energy and transport. Agriculture and land use change also emit large quantities of greenhouse gases, as does the production of cement.
Human emissions of greenhouse gases began to increase during the industrial revolution and have increased far more since the middle of the 20th century. Today, some countries have levelled off or reduced their greenhouse gas emissions. However, global emissions of greenhouse gases continue to rise.
Some greenhouse gases can also occur naturally, however emissions of these are balanced by their natural absorption. See our page on the carbon cycle for some examples of these.
Examples of greenhouse gases
The most abundant greenhouse gases are listed below. For data on the concentrations of some of these gases in the atmosphere, please view our Climate Dashboard.
- Carbon dioxide (CO2) – this is the principal greenhouse gas emitted by human activity, mostly burning fossil fuels. The concentration of CO2 in the atmosphere has increased by more than 50% since the beginning of the industrial revolution.
- Methane (CH4) – the largest human sources of methane emissions are land use change and agriculture. Methane doesn't stay in the atmosphere as long as CO2, but has a Global Warming Potential (GWP) almost 30 times higher than CO2 over 100 years.
- Nitrous oxide (N2O) – another major part of emissions from agriculture, nitrous oxide has a GWP more than 270 times greater than CO2. It also damages the ozone layer.
- Ozone (O3) – when it occurs at low altitude ozone is a damaging air pollutant, however the ozone layer in the stratosphere protects us from the Sun’s harmful ultraviolet radiation (find out more on our ozone layer webpage). Ozone is also a greenhouse gas. Its impact on surface temperatures varies around the world but is around 25% that of CO2 on average.
- Chlorofluorocarbons (CFCs) and Hydrofluorocarbons (HFCs) – these artificial compounds are used in refrigerators, air conditioning systems, aerosol sprays and cleaning agents, among other uses. HFCs are replacing CFCs, as CFCs are extremely damaging to the ozone layer and are being phased out under the Montreal Protocol. However, both are potent greenhouse gases, with GWP values ranging from several hundred to several thousand times greater than CO2. Despite this, their relatively low concentration in the atmosphere means their overall contribution to global warming is quite low compared to CO2.
- Other fluorinated compounds – as well as CFCs and HFCs, many other potent greenhouse gases contain fluorine. Examples include Nitrogen trifluoride (NF3) and Tetrafluoromethane (CF4). Another example, Sulfur hexafluoride (SF6), is the most potent known greenhouse gas, with a GWP 23,500 times greater than CO2.
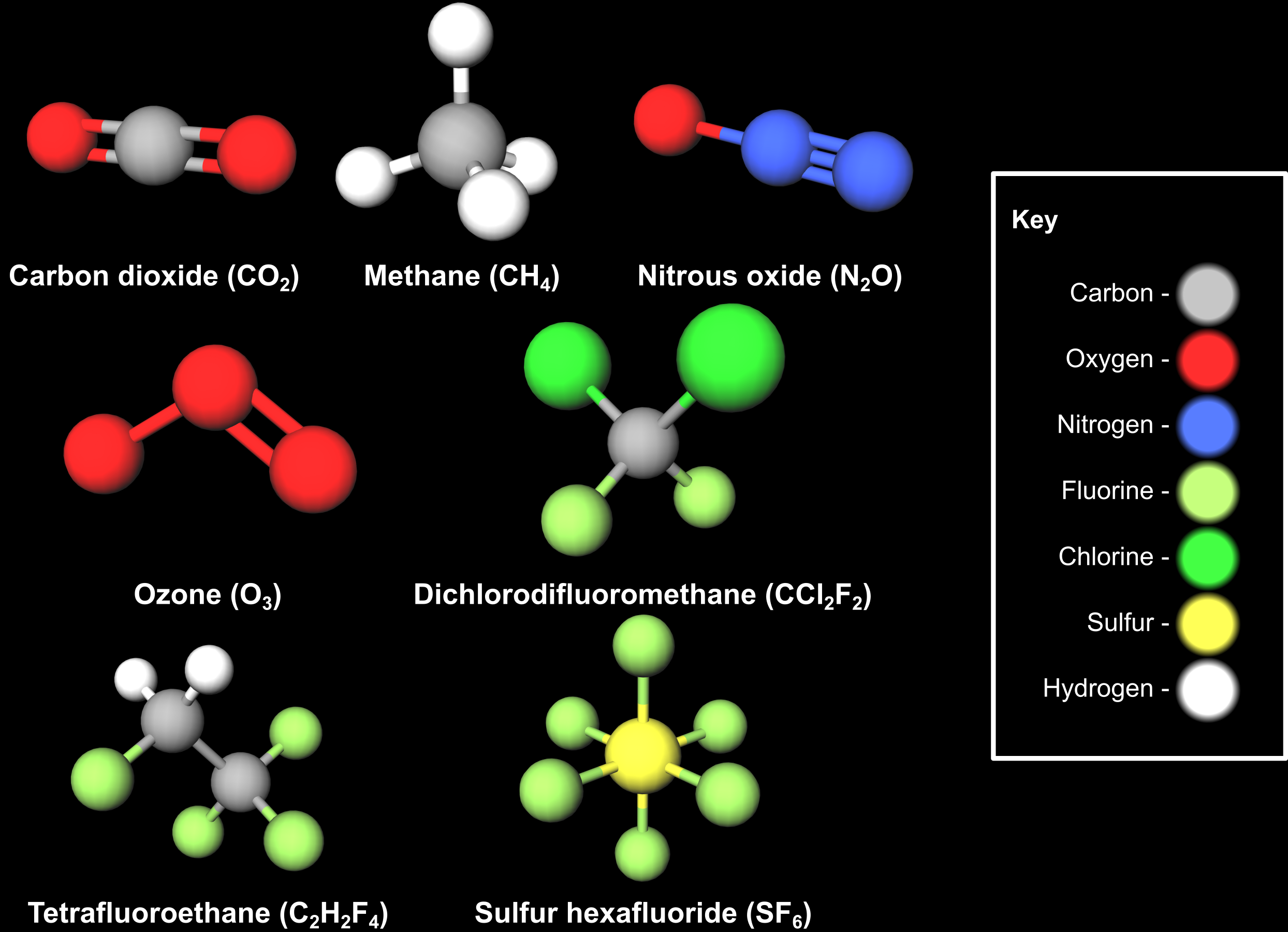
Some of the major greenhouse gases described on this page. Dichlorodifluoromethane is a chlorofluorocarbon, and Tetrafluoroethane is a hydrofluorocarbon.
Is water vapour a greenhouse gas?
Yes, water vapour is a potent greenhouse gas and is responsible for most of the overall greenhouse effect. However, the amount of water vapour in the atmosphere changes in a very different way from the other greenhouse gases described above.
Water vapour quickly reaches an equilibrium in the atmosphere according to temperature, with its level in the atmosphere either increasing by evaporating from the surface of the ocean, or falling by precipitating out as rain. This means that water vapour responds to changes in global temperature quickly and to a far greater extent than other greenhouse gases that have natural sources, which stay in the atmosphere much longer than water vapour.
A warming atmosphere can hold more water vapour and water vapour is capable of adding to warming that has been induced by some other process. However, if temperatures then fell, the amount of atmospheric water vapour would fall too.
This feedback effect means water vapour is not a driver of the global warming we are seeing today, despite its strength as a greenhouse gas. The greenhouse gases produced by human activity add to the existing greenhouse effect and cause global warming.


